What Are the Hallmarks of Aging? Insights from the Practical Anti-Aging Symposium 2026 on Advanced Cellular Health Strategies
On February 26–27, 2026, in Bangkok, the Practical Anti-Aging Symposium 2026 was held as an academic forum in anti-aging and preventive medicine, bringing together the latest insights on advanced, cell-level health optimization.
The event brought together physicians and experts in longevity medicine from various disciplines to exchange knowledge on long-term health strategies, emerging medical technologies, and the biological mechanisms underlying the aging process.
One of the participants at this symposium was Dr. Tanaporn Eiamprapai, MD, who attended the sessions and engaged in knowledge exchange on the latest concepts and research in longevity medicine and anti-aging science, with the aim of applying these insights to enhance patient care.
One of the key topics gaining significant attention in the field of anti-aging medicine today is the concept known as the “Hallmarks of Aging,” a scientific framework that helps explain the biological mechanisms underlying the aging process.
This concept was developed by scientists in biology and anti-aging medicine to better understand why the human body declines over time and what factors contribute to the development of age-related chronic diseases.
Aging is not merely a matter of chronological age, it is the result of continuously accumulating biological processes at the cellular level.
Over the past decade, the field of longevity medicine has advanced a framework that systematically explains these “mechanisms of decline,” known as the Hallmarks of Aging.
This perspective shifts how we view aging from an inevitable fate to a biological process that can be understood, measured, and potentially optimized.
What Are the Hallmarks of Aging?
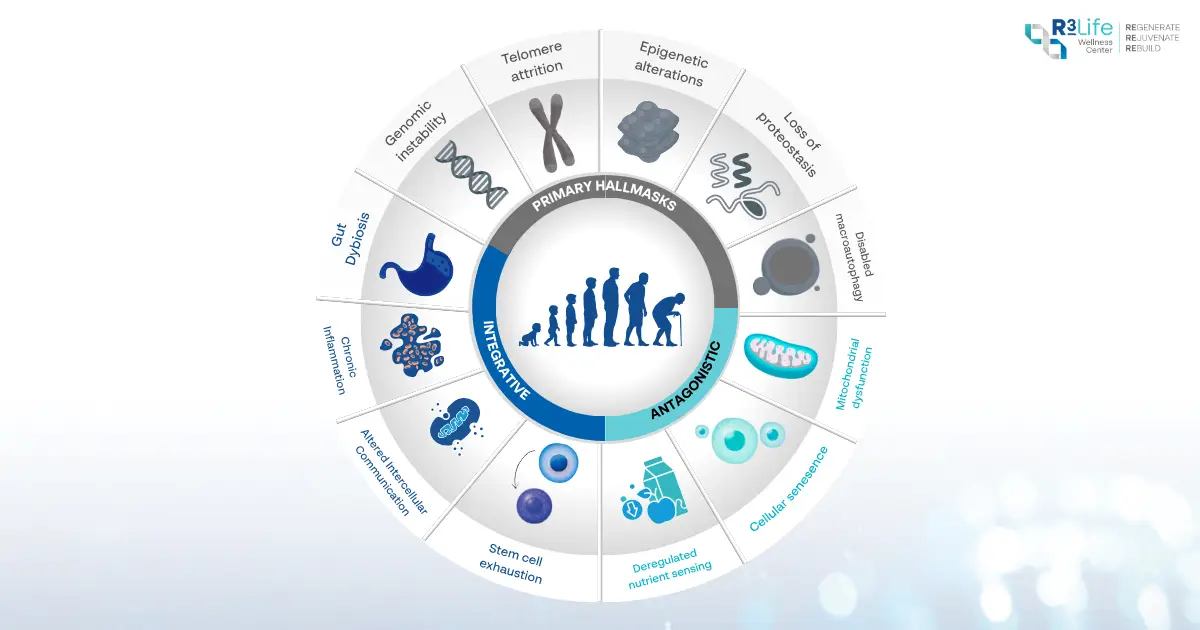
The Hallmarks of Aging were first introduced in the journal Cell in 2013 and later updated in 2023, expanding the framework to a total of 12 hallmarks (Cell, 2013; Cell, 2023).
Rather than being a list of trending terms, the Hallmarks represent a structured set of biological mechanisms. They are not a simple checklist, but a comprehensive framework that explains how cellular decline occurs and in what sequence it progresses.
The Hallmarks are categorized into three main mechanistic groups as follows:
1) Primary Hallmarks: The Initial Damage to Biological Systems
These are fundamental alterations that accumulate directly within cells, including:
- Genomic instability – DNA damage and mutations accumulate over time leading to Genomic Instability and impaired cellular function. caused by reactive oxygen species (ROS), errors during cell division, telomere dysfunction or exposure to toxins. As DNA repair mechanisms become less efficient, mutations accumulate over time.
- Telomere attrition – Telomeres are protective caps at the ends of chromosomes. With each cell division, they gradually shorten. Once they reach a critical length, cells enter a state of senescence.
- Epigenetic alterations – Changes in DNA methylation patterns and histone modifications disrupt normal gene expression and regulation.
- Loss of proteostasis – Cellular systems responsible for protein quality control, such as autophagy and the proteasome, become less effective, leading to the accumulation of misfolded or damaged proteins.
- Disabled macroautophagy – A dysfunction or failure of the cell’s internal waste-clearing and recycling system (autophagy), resulting in the buildup of defective proteins and damaged organelles.
Primary hallmarks can be thought of as the “silent cracks” that gradually form within the biological system.
2) Antagonistic Hallmarks: Protective Mechanisms That Become Drivers of Aging
- Deregulated nutrient sensing – Key pathways such as mTOR, AMPK, and insulin/IGF-1 become imbalanced, promoting continuous growth without adequate repair and maintenance.
- Mitochondrial dysfunction – ATP production declines, reactive oxygen species (ROS) increase, and mitophagy becomes less efficient, leading to cellular energy imbalance.
- Cellular senescence – Cells stop dividing but remain metabolically active, secreting pro-inflammatory factors through the SASP (senescence-associated secretory phenotype), which contributes to chronic low-grade inflammation and can suppress or dysregulate normal immune function.
These mechanisms originally serve protective roles, but when they become chronic, they accelerate the trajectory of aging.
3) Integrative Hallmarks: When Repair Systems Begin to Fail
- Stem cell exhaustion – Both the number and functional capacity of stem cells decline, reducing the body’s ability to repair and regenerate tissues.
- Altered intercellular communication – Disrupted signaling between cells, with increased inflammatory signaling and dysregulation in neuroimmune and endocrine pathways. Most-reported changes in circulating hormone concentrations and hormone profiles with aging.
- Chronic inflammation – A persistent, low-grade systemic inflammation. At this stage, aging is no longer confined to the cellular level but manifests as functional decline across organs.
- Dysbiosis – An imbalance in the body’s microbial ecosystem, particularly in the gut, where beneficial bacteria decrease and harmful microbes proliferate. This can impair digestion (e.g., bloating, diarrhea), weaken the immune system, and contribute to the development of chronic diseases.
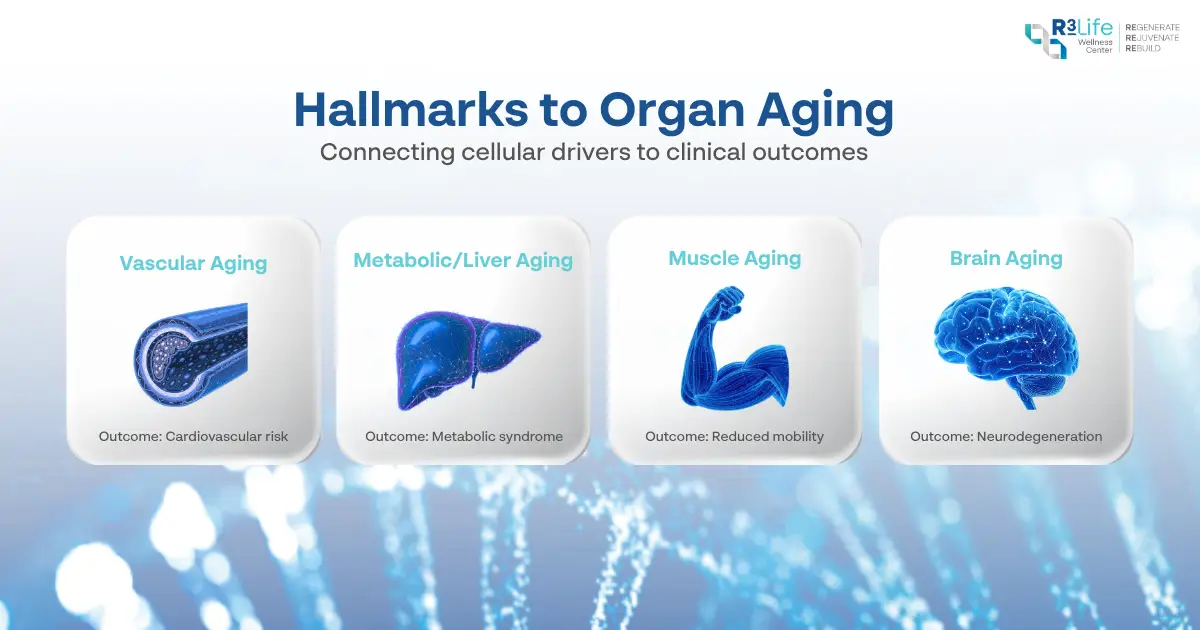
From Cellular Mechanisms to Organ Decline: When Hallmarks Become Clinical Disease
The Hallmarks of Aging do not act in isolation. They converge and interact, ultimately impacting vital organs.
This is a critical point: while most people understand diseases, far fewer understand the underlying mechanisms that drive them.
Hallmarks → Organ Aging → Clinical Disease
1. Vascular Aging: The Starting Point of Cardiovascular Disease
Cellular mechanisms:
- Mitochondrial dysfunction in endothelial cells
- Reduced nitric oxide availability
- Telomere shortening
- Cellular senescence
- NF-κB activation
These changes disrupt endothelial homeostasis, leading to arterial stiffness and endothelial dysfunction.
This represents the pathway from mitochondrial dysfunction → inflammation → vascular aging → cardiovascular disease, closely linked to research in inflammaging and vascular senescence.
2. Metabolic / Liver Aging: When Energy-Sensing Systems Become Dysregulated
When key energy-regulating hallmarks become imbalanced:
- mTOR overactivation driven by hyperinsulinemia
- Reduced autophagy
- Increased lipotoxicity
- Elevated Endoplasmic Reticulum stress and activation of inflammatory cascades
These disruptions lead to fat accumulation in the liver (NAFLD) and the development of metabolic syndrome.
NAFLD can therefore be seen as a clinical manifestation of dysregulated nutrient sensing combined with mitochondrial stress.
3. Muscle Aging: Decline in Regenerative Capacity
Satellite cells, the muscle’s resident stem cells, undergo telomere shortening and are affected by niche inflammation.
As stem cell exhaustion progresses, it leads to:
- Slower muscle repair
- Reduced ATP production
- Decreased VO₂ max
This is why muscle mass is considered a key longevity biomarker, ultimately contributing to sarcopenia and the frailty cascade.
4. Brain Aging: Where Proteostasis and Vascular Decline Converge
Cellular mechanisms:
- Reduced autophagy
- Accumulation of amyloid and misfolded proteins
- Increased neuroinflammation
- Vascular aging leading to reduced cerebral perfusion
Multiple hallmarks converge in the brain, collectively increasing the risk of cognitive decline that could lead to Alzheimer’s disease.
Lifespan vs. Healthspan vs. Peakspan: What’s the Difference?
The Hallmarks of Aging help explain why lifespan may increase, while healthspan and peakspan may actually shorten. Here’s how they differ:
- Lifespan – the total length of time a person lives
- Healthspan – the period of life spent free from chronic disease
- Peakspan – the phase during which the body and brain perform at their highest capacity
The Hallmarks of Aging serve as a framework to guide when to focus on prevention, when to prioritize detection and interception, and when to emphasize preservation and resilience, ultimately helping extend not just lifespan, but also healthspan and peakspan.

Clinical Focus Across the Lifespan: Tailoring Hallmarks of Aging at Different Life Stages
The Hallmarks of Aging do not emerge simultaneously, nor do they progress at the same rate throughout life. Therefore, aging management should not be approached the same way at age 25 as it is at 55.
Within a systems-based longevity medicine framework, strategies can be tailored across different life stages as follows:
1) Early Life: Programming Phase
From childhood through adolescence, this stage represents a critical window where epigenetic programming shapes long-term health trajectories.
Key priorities during this phase include:
- Mitochondrial health
- Balanced nutrient sensing
- Metabolic programming
Epigenetic alterations occurring early in life can influence future risk of metabolic diseases and vascular aging (as described in the Developmental Origins of Health and Disease hypothesis).
This is the stage where the hallmarks begin to be “set,” but have not yet manifested clinically.
2) Young Adulthood: Prevention Phase
In early adulthood, several hallmarks begin to accumulate silently:
- Telomere shortening starts to become evident
- Insulin resistance begins to develop
- Chronic low-grade inflammation emerges
However, overt disease is typically not yet present. This stage represents a critical window for prevention.
Managing visceral fat, optimizing sleep and circadian rhythm, and maintaining muscle mass can significantly slow the trajectory of mitochondrial dysfunction and nutrient dysregulation.
3) Midlife: Detection & Interception Phase
Between the ages of 40–60, multiple hallmarks begin to interconnect and amplify one another:
- Increased senescent cell burden
- More pronounced telomere attrition
- Rising ApoB and CRP levels
- Gradual increase in HbA1c
At this stage, biological aging may begin to outpace chronological age in some individuals. Strategies should therefore focus on: Advanced biomarker testing, Telomere length assessment, Evaluation of metabolic resilience and Vascular risk assessment. This is the phase of interception, an opportunity to intervene before overt organ dysfunction becomes clinically apparent.
4) Older Age: Preservation & Resilience Phase
Late Life: Preservation & Resilience Phase
As individuals enter older age, many hallmarks progress into the integrative phase:
- More pronounced stem cell exhaustion
- Increased sarcopenia
- Elevated chronic inflammation
At this stage, the goal is no longer to “reverse aging,” but to preserve functional reserve focusing on: Muscle preservation, Cognitive support, Vascular stability and Optimization of the regenerative environment. This approach aims to maintain healthspan, even as lifespan continues.
Strategic Longevity Medicine
The Hallmarks of Aging help us understand the underlying mechanisms, while Clinical Focus Across the Lifespan helps us understand the timing.
If we manage the hallmarks at the right stages, we can slow telomere attrition, reduce senescent burden, control metabolic dysregulation, and support the stem cell niche.
This is the fundamental difference between treating disease and designing the trajectory of life.
Telomeres: A Key Indicator of Biological Aging
Telomeres are repetitive DNA sequences that protect chromosomes from instability. With each cell division, telomeres shorten by approximately 50–200 base pairs.
Their primary role is to safeguard DNA integrity. Once telomeres reach a critical length, cells enter replicative senescence.
At this critical threshold, the following processes are activated:
- DNA damage response
- p53 pathway activation
- Replicative senescence
Factors that accelerate telomere shortening include:
- Oxidative stress
- Chronic inflammation
- Insulin resistance and type 2 diabetes
- Psychological stress
Why Are Telomeres Important in Longevity Medicine?
Because they are one of the Hallmarks of Aging that can be measured.
Although telomere length is not the sole biomarker of aging, when assessed alongside inflammatory markers and a metabolic profile, it can help reflect an individual’s biological aging trajectory.
Therefore, measuring telomere length serves as one of the tools for evaluating biological aging, though not the only one.
Many clinics, including R3 Life Wellness Center, offer telomere length testing, often referred to as assessing the body’s “current biological age” at the cellular level.
Stem Cells and Telomeres: A Deeper Biological Connection
Stem cell exhaustion is a key integrative hallmark of aging. As the stem cell niche deteriorates, telomerase activity declines, leading to reduced regenerative capacity and impaired tissue repair efficiency.
When exploring the relationship between stem cells and telomeres, the connection lies in how stem cell function can help modulate the rate of telomere attrition and support cellular longevity.
Mesenchymal stem cells (MSCs) may contribute through several mechanisms:
- Reducing chronic inflammation
- Chronic inflammation accelerates telomere shortening. MSCs can secrete anti-inflammatory cytokines, helping to mitigate this process.
- Optimizing the stem cell niche
- By improving the microenvironment, MSCs may enhance the function of resident stem cells.
- Lowering oxidative stress
- Oxidative stress is a key driver of telomere shortening. MSCs may help reduce this burden.
- Supporting repair systems
- By enhancing tissue repair and cellular maintenance, MSCs may help slow the rate of telomere loss over time.
Importantly, stem cell therapy may not directly lengthen telomeres, but it may help slow telomere attrition and improve the regenerative microenvironment.
This approach should always be personalized and clinically evaluated—stem cells are not a “miracle cure.” However, within longevity medicine, assessing both stem cell health and telomere status is part of a more integrated, systems-based strategy for optimizing long-term health.

Universal Accelerators: What Speeds Up the Hallmarks of Aging?
Key accelerating factors include:
- Visceral adiposity and insulin resistance
- Sleep disruption and circadian misalignment
- Physical inactivity and muscle loss
- Chronic psychosocial stress
- Smoking and toxic exposures
- Excessive alcohol consumption
These factors drive mitochondrial dysfunction, increase inflammation, and promote cellular senescence at a faster rate.
The result is “accelerated biological aging” where biological age exceeds chronological age.
Conclusion
The Hallmarks of Aging are not merely a way to describe aging—they serve as a map of cellular decline, helping identify where meaningful interventions can take place.
When we assess:
- Telomere length
- Inflammatory load
- Metabolic resilience
- Muscle reserve
- Vascular markers
We shift our approach from treating end-stage disease to managing the upstream drivers of biological aging.
The real question is no longer “How old am I?” but rather “In which direction is my biology heading?”
By evaluating telomere length, inflammatory burden, metabolic resilience, and muscle reserve, we are systematically assessing both healthspan and peakspan.


 How Heavy Metal Buildup Quietly Accelerates Aging — And What You Can Do About It
How Heavy Metal Buildup Quietly Accelerates Aging — And What You Can Do About It
 Vitamin D3 Injection: The Power for Boost Immunity & Anti-Aging Naturally
Vitamin D3 Injection: The Power for Boost Immunity & Anti-Aging Naturally




